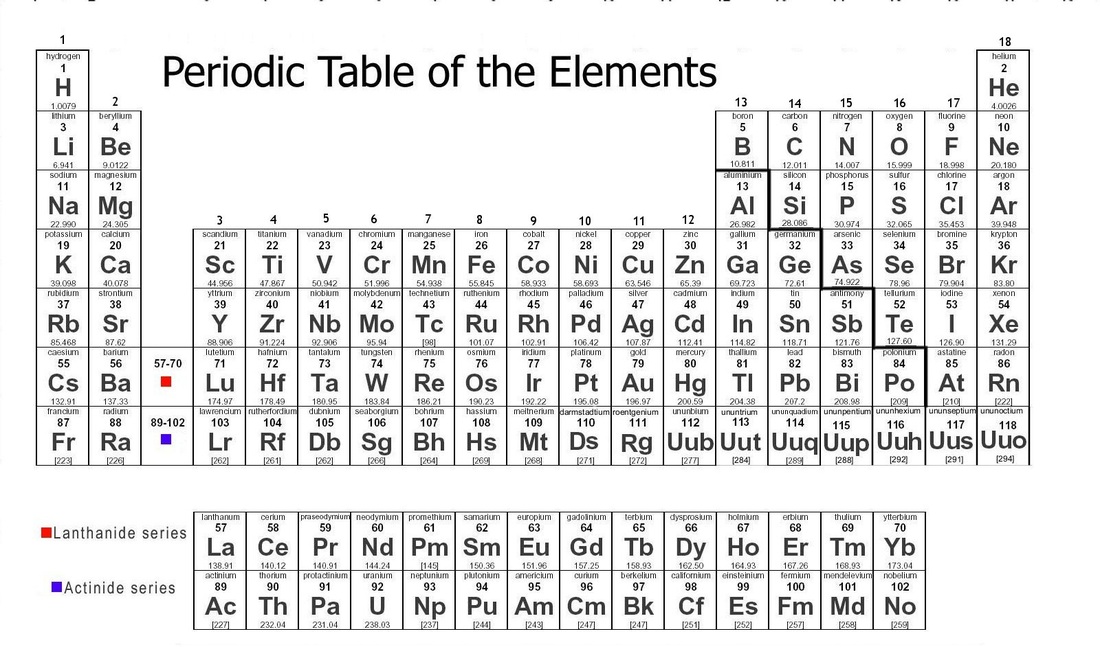
Periodic Table
Periods:
Periods are the rows in the Periodic table. They tell us how many energy levels the element has in that row.
Groups (Families):
Groups, or families, are the columns in a Periodic table. The tell us how many valence electrons the element in that column has. The first and second columns have one and two valence electrons. You skip the transition metals because they all have 2 valence electrons. The Boron column has 3 valence electrons and it continues on till the 8th column. Helium has 2 valence electrons, but from neon down those elements have 8 valence electrons.
How are the elements in the Periodic Table classified?
Elements in the periodic table are in order by their atomic number, or the number of protons they have.
Valence Electrons:
Valence electrons are the electrons on the outer ring of the electron cloud. They are determined by the group number they are in. The only exception are the transition metals, which all have 2 valence electrons.
Metals:
Nonmetals:
Nonmetals are located in the top right corner of the Periodic Table.
Metalloids:
Metalloids are found in the middle of the metals and nonmetals. All the elements that touch the zig-zag line on the Periodic Table are metalloids except for Aluminum.
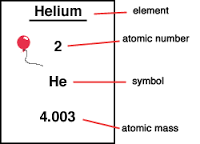
Atomic NumberThe atomic number is the number of protons and, in the case of a stable element, the number of electrons. This is how the Periodic Table is arranged. They are found at the top of the rectangle
|
Atomic Mass:The atomic Mass of an element is the total number of protons and neutrons in the element, since both of them are the only sub atomic number in the atoms that has a mass. You always round up when determining the atomic mass. The atomic mass is located at the bottom of the rectangle.
|
Reactive or not?
You can determine if an element if reactive or not by seeing how close to the center of the table it is. Group 1 and Group 17 are the most reactive and the closer you get in the less reactive the element gets. Group 18 is special though. They are called Noble Gases, which means they are not reactive at all. An element is more reactive the closer it gets to a completed outer ring, which mean 8 electrons in the outer ring. Once it reaches 8 electrons, like Group 18 has, it is no longer reactive, since it needs no more electrons.
Properties of Metal
Metals have very unique qualities. They are generally shiny, malleable, and hard. They are also conductors.
Properties of Non-metals
Non-metals can conduct heat or energy, but not very well. They are usually brittle and cannot be molded into shapes easily.
Properties of Metalloids
Metalloids have the characteristics of both metals and non-metals. They are semi-conductors , which means they can insulate and conduct energy well.